Western blot Research Application Note:
EMRE Is an Essential Component of the Mitochondrial Calcium Uniporter Complex
Article in Science 342(6164) · November 2013 with 54 Reads
Yasemin Sancak, Andrew L. Markhard, Toshimori Kitami, Erika Kovács-Bogdán, Kimberli J. Kamer, Namrata D. Udeshi, Steven A. Carr, Dipayan Chaudhuri., David E. Clapham, Andrew A. Li, Sarah E. Calvo, Olga Goldberger, Vamsi K. Mootha
DOI: 10.1126/science.1242993 · Source: PubMed
Abstract
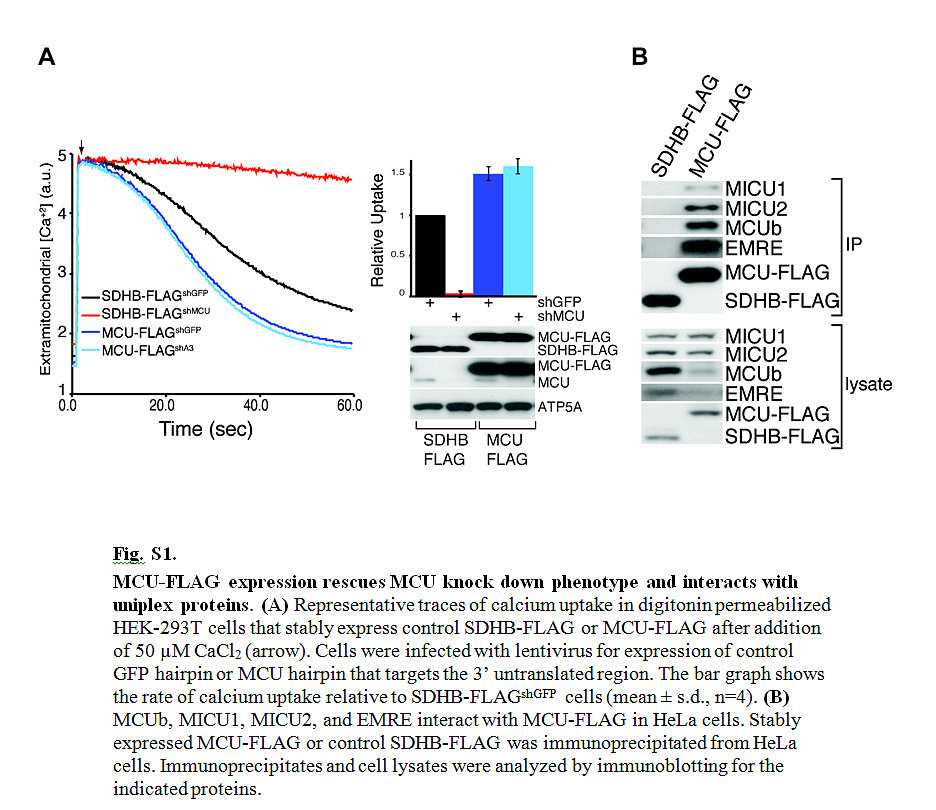
The mitochondrial uniporter is a highly selective calcium channel in the organelle’s inner membrane. Its molecular components include the EF-hand–containing calcium-binding proteins mitochondrial calcium uptake 1 (MICU1) and MICU2 and the pore-forming subunit mitochondrial calcium uniporter (MCU). We sought to achieve a full molecular characterization of the uniporter holocomplex (uniplex). Quantitative mass spectrometry of affinity-purified uniplex recovered MICU1 and MICU2, MCU and its paralog MCUb, and essential MCU regulator (EMRE), a previously uncharacterized protein. EMRE is a 10-kilodalton, metazoan-specific protein with a single transmembrane domain. In its absence, uniporter channel activity was lost despite intact MCU expression and oligomerization. EMRE was required for the interaction of MCU with MICU1 and MICU2. Hence, EMRE is essential for in vivo uniporter current and additionally bridges the calcium-sensing role of MICU1 and MICU2 with the calcium-conducting role of MCU.
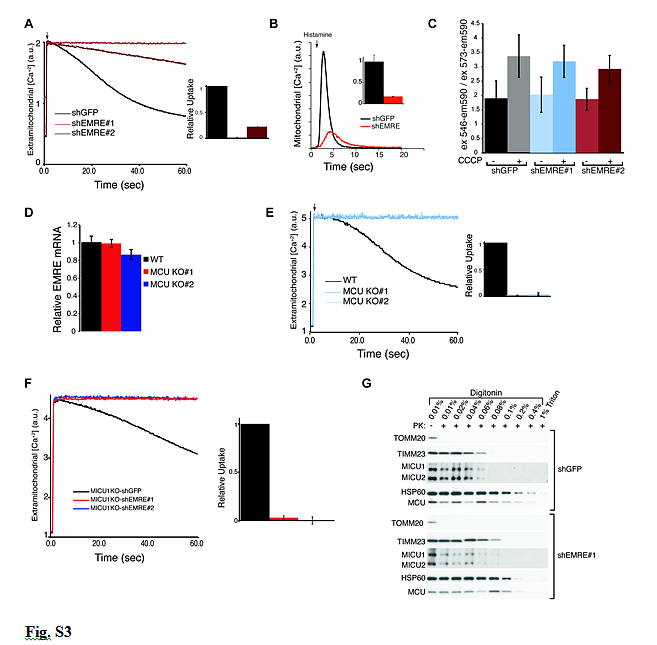
Figure S3 Discussion
-
- Loss of EMRE impairs uniporter uptake despite normal mitochondrial membrane potential and proper submitochondrial localization of uniplex proteins.
- (A) EMRE knock down impairs mitochondrial calcium uptake in HeLa cells. Representative traces of calcium uptake in digitonin permeabilized HeLa cells with GFP (control) and EMRE knock down after addition of 50 µM CaCl2 (arrow). The bar graph shows the rate of calcium uptake relative to shGFP cells (mean ± s.d., n=3). (B) EMRE knock down impairs mitochondrial calcium uptake in intact HeLa cells as measured by mt-Aequorin after histamine stimulation (arrow). Intact cells that express mt-Aequorin were stimulated with histamine (arrow). Increase in mitochondrial calcium was monitored using luminescence. Averaged traces of four experiments from control (shGFP) and EMRE knockdown are shown. Inset shows relative upake in EMRE knockdown cells compared to control (mean ± s.d., n=4). (C) EMRE knock down does not alter mitochondrial membrane potential. shEMRE and control shGFP knock down HEK-293T cells were incubated with TMRM in the absence and presence of CCCP.
Materials
Reagents were obtained from the following sources: MICU2 antibody from Abcam (ab101465), EMRE antibody from Santa Cruz Biotechnology (sc-86337), anti-FLAG M2 affinity gel from Sigma (A2220), FLAG peptide from Sigma (F3290), rabbit FLAG antibody from Cell Signaling Technology (2368S), MCU antibody from Sigma (HPA016480), TOMM20 antibody from Santa Cruz Biotechnology (sc17764), TIMM23 antibody from BD Bioscience (611222), HSP60 antibody from Abcam (ab3080), MCUb antibody from Abgent (AP12355B), ATP5A antibody from Abcam (ab14748), DDM from Sigma (D4641), digitonin from Sigma (D141), PMSF from Sigma ( P7626), Oregon Green® 488 BAPTA-6F from Life Technologies (O23990), NativePAGE Novex 3-12% Bis-Tris gels from Life Technologies (BN1003BOX), micro biospin chromatograpy columns from Bio-Rad (732-6204), protease inhibitor tablets from Roche Diagnostics (05056489001). MCU antibody was raised in chicken against full length human MCU. MICU1 antibody was raised in rabbit against human MICU1. For western blotting, membranes were incubated with the primary antibodies in 5% milk in PBST overnight at 4˚C, subsequent washes and secondary antibody incubations were done at room temperature using a BlotCycler™ (Precision Biosystems). Secondary antibodies were diluted 1:5000 in 5% milk in PBST.